
MHRA Guidance Note 6
MHRA Guidance Note 6 – Notes for applicants and holders of a Wholesale Dealer’s Licence (WDA(H)) or Broker Registration was updated in November 2022 with some significant changes regarding Brexit.

MHRA Guidance Note 6 – Notes for applicants and holders of a Wholesale Dealer’s Licence (WDA(H)) or Broker Registration was updated in November 2022 with some significant changes regarding Brexit.
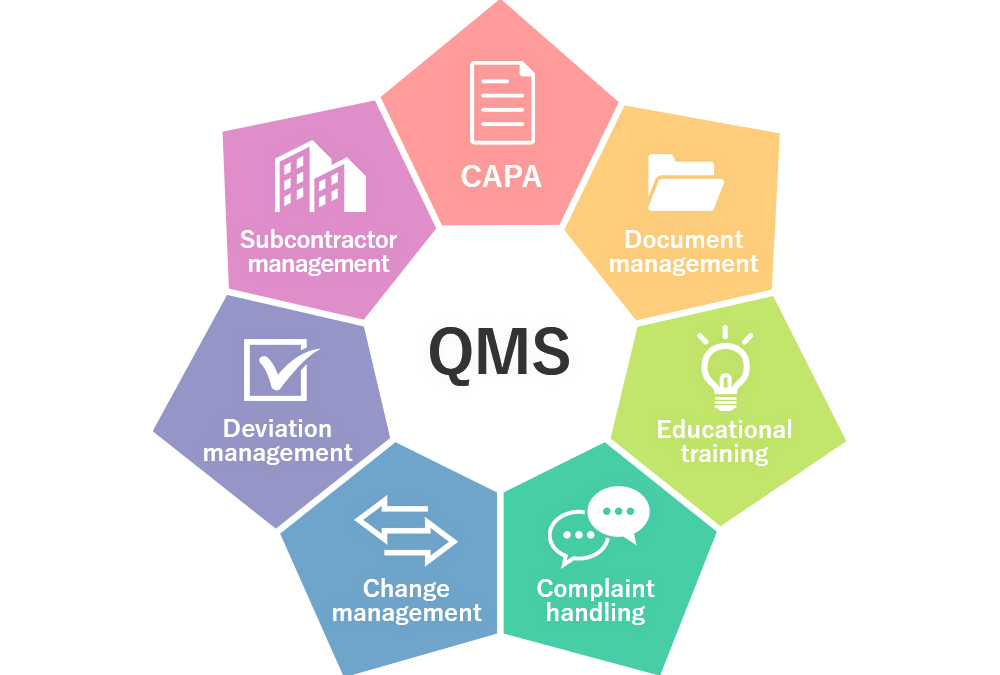
Robust Quality Management Systems (QMS) are the heart of our Quality Management. With the right process in place for your business model and the right people managing it, the Quality System will be robust, compliant, easy to navigate and a fantastic tool for trending.

Is your scope of Wholesale Distribution Authorisation (WDA) out of date? – Are you aware that the Wholesale Distribution Authorisation (WDA) categories have changed?

Licences for Medicines Wholesale – Do you know what is required by you as a pharmaceutical wholesaler to fulfil legal requirements when working with medicines? Read on to learn the basics of why you need a WDA and where to find further information to accelerate your learning.
